What is a Light Emitting Diode?
| Previous Topic | Return to OLED Menu | Next Topic |
p-type and n-type Materials
Inorganic Light emitting diodes have become very common in the last decade, used in everything from traffic lights to taillights. Inside these devices there is a very small inorganic multi-layer diode material that is driven in forward bias. Light is emitted isotropically, and a cone is used to focus the light in a single direction. In the last decade, inexpensive green and blue LEDs have become readily available in addition to the more common red.
Simplified structure of a diode:
- A piece of p-type semiconductor material is created from silicon with excess dopants such as boron.
- An n-type semiconductor is created from silicon doped with phosphorous.
- Joining the two pieces forms a p-n diode, and creates the the potential for charge transfer between the two materials.
- The n part of interfacial area of electrons is depleted
- The p part of the interface of holes is depleted.
- This creates a built-in potential, Vbi.
Equilibrium in Diodes
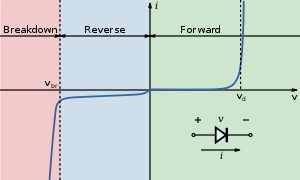
Diodes provide rectification. Current only flows in one direction. When voltage is applied and current passes through a resistor you get a linear plot known as Ohms law. When current passes through a diode you get an asymmetry in the voltage current response. Increasing the positive bias causes an exponential increase in the current. Conversely, with a reverse bias, the current flow decreases. A good diode should have very low turn-on voltage (Vd) with exponential increase, and very low current with negative bias.
The Shockley equation describes the current through the diode at any bias.
- <math>j= j_s \left ( \frac {qV_A} {e^nkT} \right ) -1\,\!</math>
see wikipedia:Diode#Shockley_diode_equation.
Applying voltage results in current flowing across the depletion layer region.
p-n diode simulation
<swf width="600" height="500">http://depts.washington.edu/cmditr/media/pndiode.swf</swf>
This is an energy level diagram as a function of distance across a PN diode. A condensed phase system is at equilibrium when the Fermi level in both the N region and the P region are exactly the same. The Fermi level is the average energy level of an electron entering or leaving the solid. There is significant energy difference between the N region and the P region for both electrons and holes because of the depletion of majority carriers that has occurred in that system.
Reverse Bias in Diodes
Reverse biased diodes have even more significant depletion. The built-in voltage increases by Vd, and the depletion region thickness increases. Very little current will flow in purified materials. In reverse bias the Fermi levels are no longer aligned because the system is not in equilibrium. There is an energy barrier that makes it very difficult for electrons in the N region to transit to the P region, and it is difficult for holes to move from the P region to the N region. This is exactly the region in a organic photovoltaic cell where the application of light causes energy absorption and causes the charge to move. However in LEDs or OLEDs the current should not flow when there is reverse bias.
Forward Bias in Diodes
In the forward biased diode, current flows and the depletion region is narrowed or eliminated. Majority carriers move from one region to the other. The energy bands are not in equilibrium, but the energy level of the P region is lower than the N region. Electrons move from the N region to the P region and holes move from P region to N region.
Forward bias in an LED moves the electrons and holes. Recombination events occur at the junction and excess free energy is emitted as light from the center region. The color of the light from an inorganic diode is controlled by the band gap energy of the semiconductors. The first LED developed was gallium arsenide, which has a low bandgap that gives the red light of early calculators. As scientists began to tailor the bandgap of 3-5 semiconductors they have achieved orange, green and most recently blue color emission.
<swf width="600" height="500">http://depts.washington.edu/cmditr/media/led.swf</swf>
| Previous Topic | Return to OLED Menu | Next Topic |