Electron Spin Resonance (ESR)
| Return to Research Tool Menu |
Background
Electron Spin Resonance (ESR) also known as Electron Paramagnetic Resonance (EPR) spectroscopy or electron paramagnetic resonance spectroscopy is a technique for studying chemical species that have one or more unpaired electrons such as free radicals, odd electron molecules, transition metal complexes, lanthanide ions, and triplet state molecules. It is analogous to NMR except it deals with spins of electrons rather than the spins of atomic nuclei. It is most useful in studying free radicals or organic complexes having transition metal ions since most stable molecules have all their electrons paired. Most ESR spectra are recorded and published as first derivatives of the original signal.
Significance
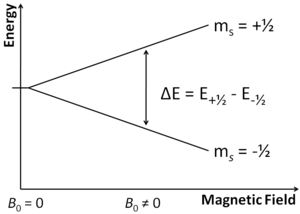
In a standard ESR experiment, the sample is irradiated with the microwave field at the constant frequency (~10GHz), the external magnetic field is slowly changed. Electron spins align with the field, but also can precess around the equilibrium orientation
The frequency of the spin precession depends on the field, it increases (linearly in a simple case) with the field increasing. When the field passes through the point, where the spin precession frequency corresponds to the microwave frequency applied, the sample will absorb microwave radiation.
In pulsed ESR a strong burst of microwave energy is applied and a complex response signal or transient signal is recorded. This is analyzed through fourier transform to yield an ESR spectrum in the frequency domain.
<swf width="500" height="400">images/2/21/Esr.swf</swf>
Operation
<embed_document width="100%" height="400">images/6/68/Esr.pdf</embed_document>