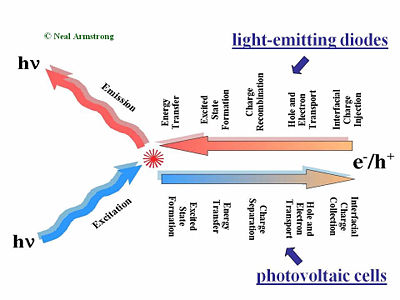
Major Processes in Organic Solar Cells
Edmond Becquerel (1820-1891) was first to discover the photovoltaic effect. But it took quite a long time before there was a report on an efficient cell and that came from Chapin, Fuller and Pearson (1954) in Bell Labs. They reported a silicon cell with 6% efficiency.
This slide gives a comparison of the processes that are placed in LED in a solar cell and in a field-effect transistor. The first step of the process required charge injections in the electrodes, holes injected from the anode, and electrons injected from the cathode. Those holes and electrons then migrate to diffuse in the presence of an external electric field due to the applied voltage across the organic layer or layers. Then the hole and the electron have to find one another to recombine. That leads to the formation of an excited site, an exciton. During its lifetime, the exciton can also hop from chain segment to chain segment or molecule to molecule. This is called an energy transfer. If all goes well, a decay to the ground state will occur radiatively and a photon will be released. This step is necessary; photons that are produced within the device must be released to see any visual information. A photovoltaic cell works in reverse to a light-emitting diode. While in a light emitting diode an electrical current is injected into the material and transformed into photons, a photovoltaic cell requires an absorption of the photons to generate an electrical current. The photovoltaic cell first absorbs photons, which leads to a formation of excitons in an excited state. These excitons move to the interface between the donor component and the accepter component in the organic solar cell. At the interface of the donor and accepter, the exciton disassociates so that the hole and the electron become free from one another. The separated hole and electron will be diffused within the material and get collected by the electrodes. The major processes occur in reverse order but there is one major difference; any organic material, or combination of materials that’s good for luminescence and thus good for light emitting diode will be bad for organic photovoltaic cells. The reason is that in a light-emitting diode, an electron and the hole approaching each other is favorable. This will form the exciton that will decay and lead to photon emission. On the other hand in the case of a photovoltaic cell, separation of the electron and hole is favorable so they can migrate to the opposing electrodes. These two steps are entirely different in the two types of systems.